|
Since neutrons have no charge, they do notinteract with atomic electrons. They are classified according totheir energies as cold, thermal, slow, intermediate, and fastneutrons. However, neutron radiationis not nearly so well understood, largely due to the fact thatneutrons have no electrical charge. and solar cosmic rays, X-rays, and gammarays as well as the alpha particles, protons, and electrons in theVan Allen belts are relatively well known. One of the more subtle of the nuclearparticles, and one of the more difficult to detect, is the neutron.Space radiation in the form of galactic.Ī beam of neutrons is attenuated whilepassing through material because the particles are either scatteredor absorbed.  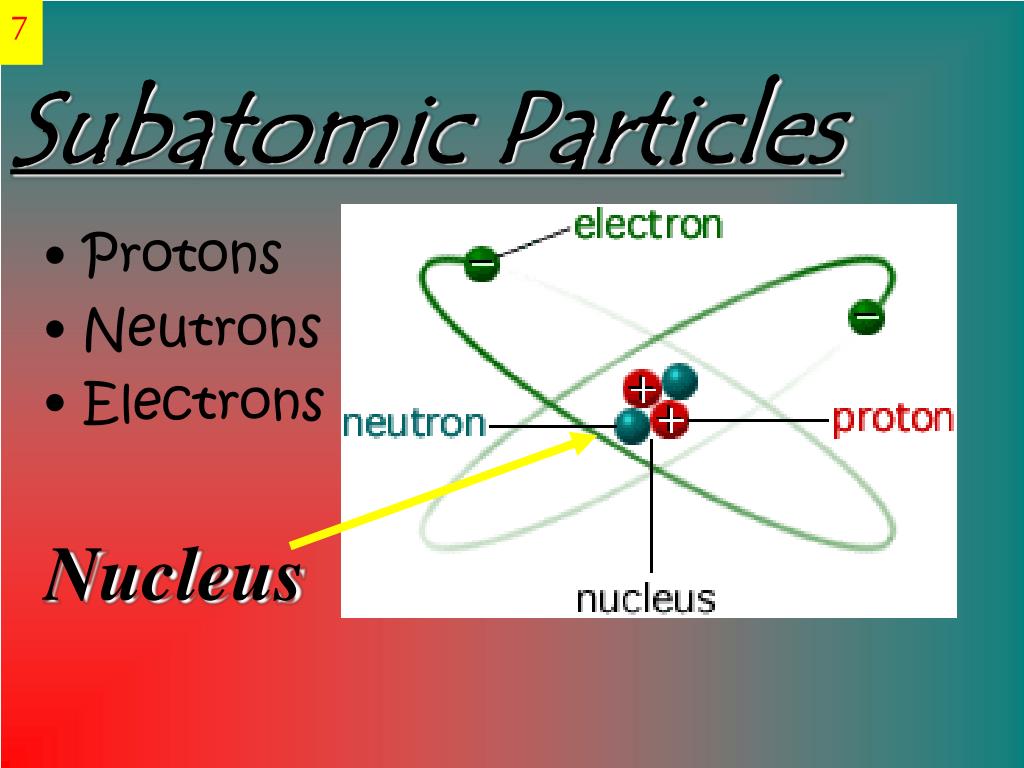
Some subatomic particles result fromnatural radioactivity when atoms with relatively unstable nucleispontaneously radiate energy or matter. Among these are electrons, protons, neutrons, alphas,and higher mass particles resulting from the processes that takeplace in the Sun and stars. Some subatomic particles exist in a free formin outer space. Since then some 200 "particles" have been found tooriginate in the nucleus! He also revealed the existence ofthe meson in 1936. The question then arises, "What is the force thatholds the particles of the nucleus together?" This force has been thesubject of investigation by modern physicists since Carl Andersondiscovered the positron in 1932. The force which binds atoms together into molecules iselectrical, as is the force that holds the atom's electrons in orbitabout the nucleus. It is the manner inwhich atoms stick together that determines the structure of themolecules. Matter is fundamentally molecular rather thanatomic, but the molecules are made up of atoms. 
Thus, atoms differ from one another in theirmass according to the number of protons and neutrons in theirnuclei. Thenucleus consists of protons and neutrons and carries a positiveelectrical charge balancing the negative charge of the electronssurrounding the nucleus. It is now called the periodic chart or table.Įarly in the 20th century, it was shown thatthe nucleus of an atom contains the vast majority of its mass. In 1868, Mendeleev, a Russian, prepared a chart of the thenknown elements, classifying them in the order of increasing atomicmasses. An atom's mass, therefore, is a characteristic of anelement. :max_bytes(150000):strip_icc()/atom-157646042-584ee6bb5f9b58a8cd2fcb02.jpg)
All atoms of a givenelement have the same mass but differ in mass from atoms of otherelements. In 1808, hetheorized that elements are composed of atoms. It was the Englishman John Dalton, however,who put the concept of an element on a firm foundation. He proposed the idea of elements, which he described as being"certain primitive and simple or perfectly unmingled bodies whichnot being made of any other bodies, or of one another, are theingredients of which all those called perfectly mixed bodies areimmediately compounded, and into which they are ultimatelyresolved."īy 1789, the French chemist Lavoisier hadlisted 23 known elements. Some of the earliest systematicinvestigations into the nature of matter were made by Robert Boyle in1661. The sciences of chemistry and physics,evolving over the years, have established a remarkable similarity tothese primitive concepts. The medieval alchemist believed that all kinds of matter had acommon origin, that they possessed one permanent "soul" housed in avariety of temporary bodies, and that these bodies could betransmuted, i.e., converted from one element to another. Man has beenprobing the nature of matter since the ancient Greeks hypothesizedthat everything is made of four elements: fire, air, water, andearth.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
